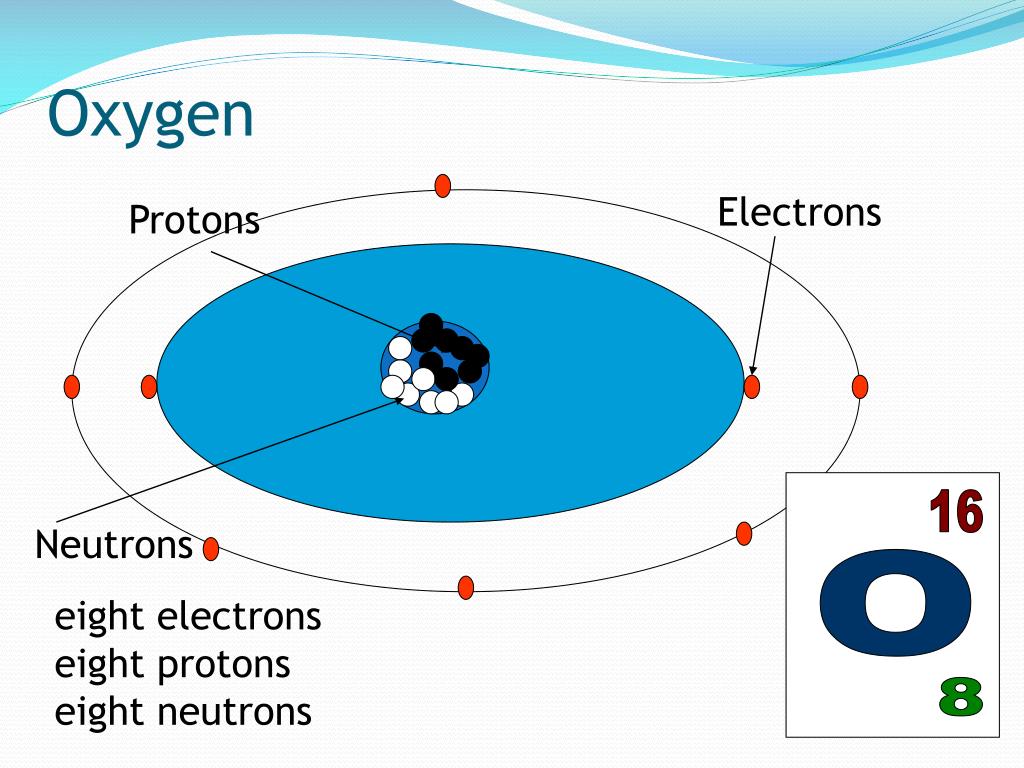
How many electrons does an oxygen atom have?Įlectrons are the permanent core particles of an atom. Therefore, an oxygen atom contains eight protons. We must always remember that the atomic number and the number of protons of an element are equal. Since oxygen is the 8th element of the periodic table, the atomic number of oxygen is 8. The elements in the periodic table are arranged according to their atomic number. There are 118 elements in the periodic table and the 8th of these elements is oxygen. The diameter of a proton particle is about 2.4 × 10 −13 cm. Its actual charge is +1.602 × 10 −19 coulombs. That is, the mass of a proton is approximately 1837 times greater than the mass of an electron. However, the actual mass of the proton is 1.6726 × 10 −27 kg. The relative mass of protons is 1, which is approximately equal to the mass of hydrogen (1.00757 amu). When a hydrogen atom removes an electron from its orbit, the positively charged particle that remains is called proton. It resides in the center or nucleus of the atom. Protons are the permanent core particles of an atom. Therefore, the number of negatively charged electrons orbiting in its orbit is equal to the number of positively charged protons in the nucleus.Ītomic number (Z) = Number of charges in the nucleus (p) How many protons does an oxygen atom have? That is, the atomic number is the total number of protons. We know that protons are located in the nucleus of an atom as a positive charge.
#OXYGEN ATOMIC NUMBER SERIAL NUMBER#
This number is equal to the serial number of the periodic table.

The atomic number of the element is expressed by ‘Z’. Thus, the number of positive charges present in the nucleus of an element is called the atomic number of that element. He called that number the order of the atoms. The results of his experiments show that each element has a unique integer equal to the number of positive charges in the nucleus of that element.

Scientist Henry Gwynn Jefferies Mosle examined the X-ray spectrum of various elements in 1913-to 1914.
#OXYGEN ATOMIC NUMBER HOW TO#
How to easily find the number of electrons, protons and neutrons in an oxygen atom? Electrons revolve around the nucleus in a specific orbit. The only exception is hydrogen, which has only protons in its nucleus but no neutrons. Experiments by various scientists have shown that the nucleus of an atom contains protons and neutrons. One is the nucleus and the other is the orbit. Also, neutrino, antineutrino, positron, and mason are located in an atom as temporary particles.Ītoms can usually be divided into two parts. Electrons, protons, and neutrons are located in the atom as permanent particles. Numerous permanent and temporary particles exist in the atom. However, it has been possible to detect atoms by increasing the vision of a very powerful electron microscope by two million times. Hopefully, after reading this article you will know the details about this topic. Also discussed is the position of electrons, protons, and neutrons in an atom, the number of atomic masses, and the isotopes of oxygen. This article discussed in detail how to easily find the number of protons, neutrons, and electrons in an oxygen atom. The oxygen atom has three stable isotopes. The number of neutrons depends on the isotope of the element. Therefore, an oxygen atom has eight neutrons. The difference between the mass number of the oxygen atom and the number of protons is eight. The number of neutrons in an atom can be determined by the difference between the atomic mass and the number of protons. Therefore, an oxygen atom has eight protons and eight electrons. The atomic number of an element is equal to the number of protons and electrons in that element. Oxygen is the 8th element of the periodic table so its atomic number is 8. Oxygen is a classified nonmetallic element and its symbol is O.
